Drug Impurities Reference Standards
Showing 21–30 of 1775 results
-
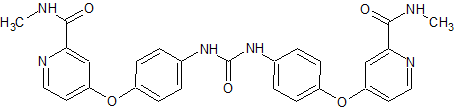
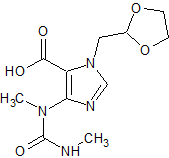
Sorafenib Impurity 3
- Product Number CT-01110-412
- Parent Drug Sorafenib
- CAS Number 284670-98-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
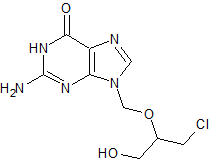
Ganciclovir EP Impurity C
- Product Number CT-01110-73
- Parent Drug Ganciclovir
- CAS Number 108436-36-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
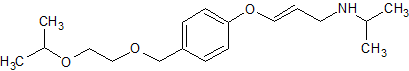
Bisoprolol EP Impurity E
- Product Number CT-01110-595
- Parent Drug Bisoprolol
- CAS Number 1217245-60-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
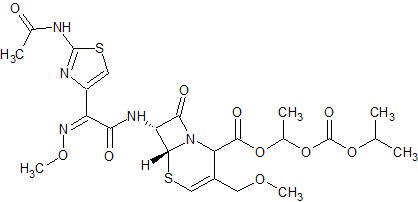
Cefpodoxime Proxetil EP Impurity G
- Product Number CT-01110-880
- Parent Drug Cefpodoxime
- CAS Number 947692-15-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Doxofylline Impurity 4
- Product Number CT-01110-865
- Parent Drug Doxofylline
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
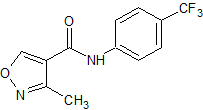
Flumazenil EP Impurity D
- Product Number CT-01110-787
- Parent Drug Flumazenil
- CAS Number 78755-80-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Indacaterol Impurity 4
- Product Number CT-01110-852
- Parent Drug Indacaterol
- CAS Number 15450-76-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
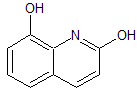
Leflunomide EP Impurity E
- Product Number CT-01110-913
- Parent Drug Leflunomide
- CAS Number 208401-20-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
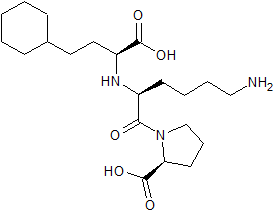
Lisinopril EP Impurity F
- Product Number CT-01110-935
- Parent Drug Lisinopril
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
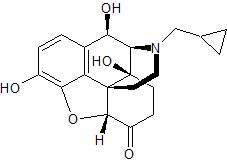
Naltrexone EP Impurity G
- Product Number CT-01110-832
- Parent Drug Naltrexone
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options