Drug Impurities Reference Standards
Showing 461–470 of 1927 results
-
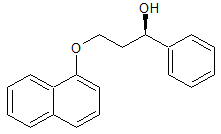
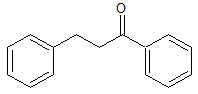
Dapoxetine Impurity 19
- Product Number CT-01110-795
- Parent Drug Dapoxetine
- CAS Number 156453-53-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
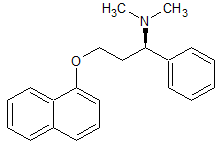
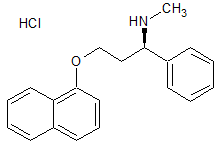
Dapoxetine Impurity 29
- Product Number CT-01110-796
- Parent Drug Dapoxetine
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
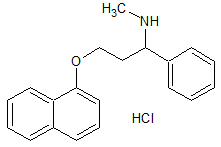
Dapoxetine Impurity 3
- Product Number CT-01110-797
- Parent Drug Dapoxetine
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
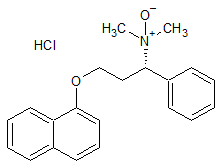
Dapoxetine Impurity 30
- Product Number CT-01110-798
- Parent Drug Dapoxetine
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Dapoxetine Impurity 34
- Product Number CT-01110-799
- Parent Drug Dapoxetine
- CAS Number 1083-30-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Dapoxetine Impurity 35
- Product Number CT-01110-800
- Parent Drug Dapoxetine
- CAS Number 1202160-36-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
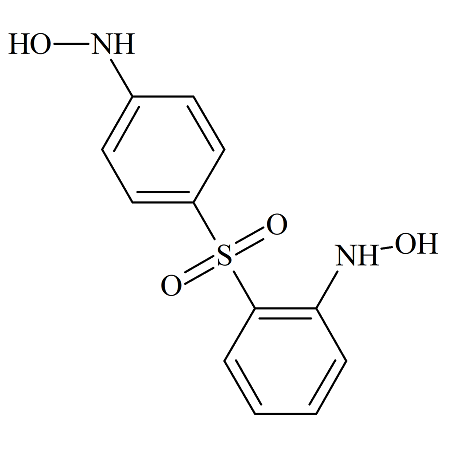
Dapsone 2,4′-dihydroxyamino Impurity
- Product Number DAP-14-003
- Parent Drug Dapsone
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
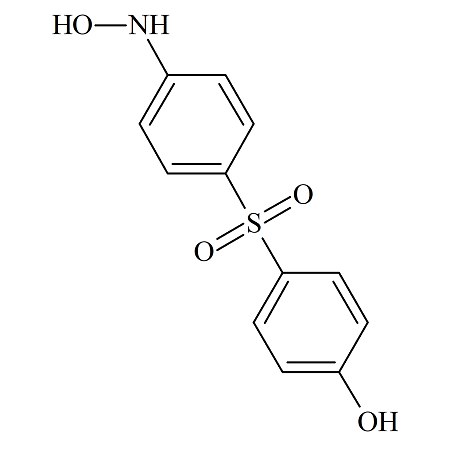
Dapsone 4-Hydroxyamino Impurity
- Product Number DAP-14-002
- Parent Drug Dapsone
- CAS Number 872811-58-0
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
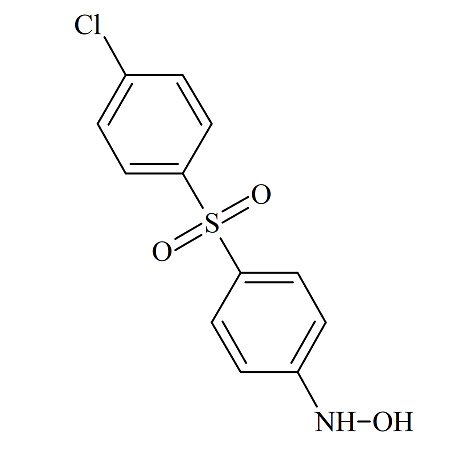
Dapsone 4-Hydroxyamino-4′-chloro Impurity
- Product Number DAP-14-004
- Parent Drug Dapsone
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
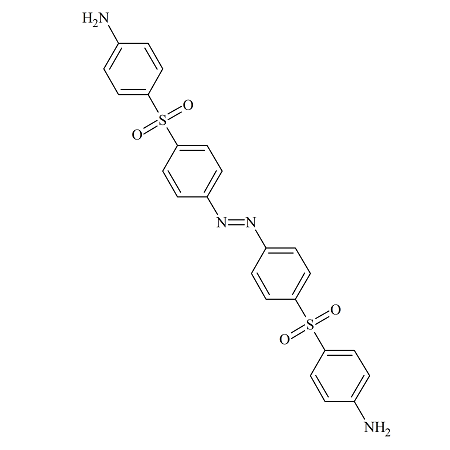
Dapsone Azo Dimer
- Product Number DAP-15-003
- Parent Drug Dapsone
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 4 to 6 week(s)See more size options