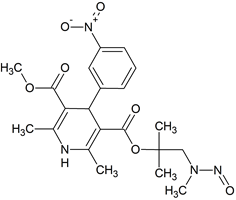Drug Impurities Reference Standards
Showing 1101–1110 of 1775 results
-
Candesartan Cilexetil EP Impurity A
- Product Number CT-01110-557
- Parent Drug Candesartan
- CAS Number 139481-58-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
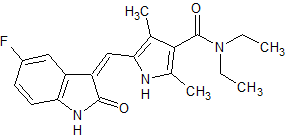
Sunitinib Impurity 25
- Product Number CT-01110-646
- Parent Drug Sunitinib
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
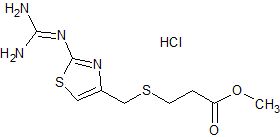
Famotidine EP Impurity J HCl
- Product Number CT-01110-722
- Parent Drug Famotidine
- CAS Number 1798006-30-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
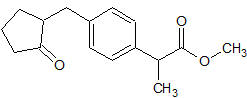
Loxoprofen Impurity 11
- Product Number CT-01110-836
- Parent Drug Loxoprofen
- CAS Number 81762-92-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
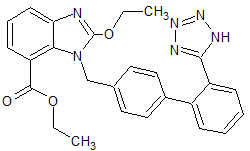
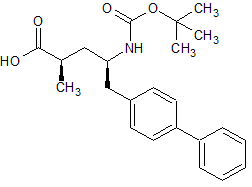
LCZ-696 Impurity 18(S,R)
- Product Number CT-01110-942
- Parent Drug LCZ-696
- CAS Number 1012341-54-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
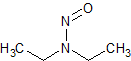
N-nitrosodiethylamine (NDEA)
- Product Number N-10521-03
- Parent Drug Nitroso Compounds
- CAS Number 55-18-5
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 2 to 3 week(s)See more size options -
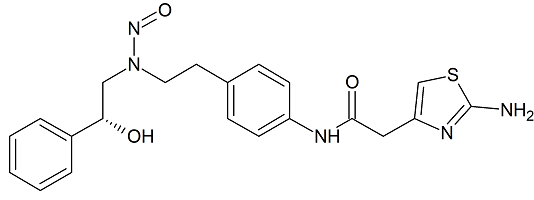
Mirabegron N-Nitroso
- Product Number M-20224-01
- Parent Drug Mirabegron
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: week(s)See more size options -
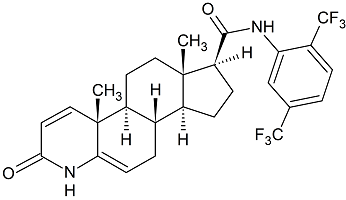
N-Des(3,3-diphenylpropyl) Lercanidipine N-nitroso
- Product Number L-31012-01
- Parent Drug Lercanidipine
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
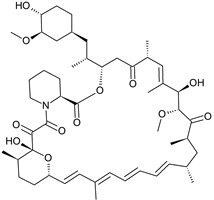
Rapamycin Tetraene Impurity
- Product Number R-41213-01
- Parent Drug Rapamycin
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options