Drug Impurities Reference Standards
Showing 1151–1160 of 1775 results
-
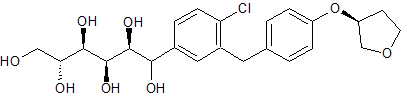
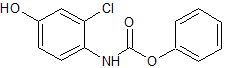
Empagliflozin Impurity 12
- Product Number CT-01110-151
- Parent Drug Empagliflozin
- CAS Number 1620758-34-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
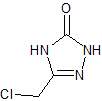
Apremilast impurity 17
- Product Number CT-01110-20
- Parent Drug Apremilast
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
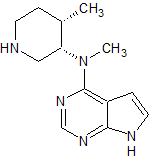
Tofacitinib Impurity 72
- Product Number CT-01110-246
- Parent Drug Tofacitinib
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
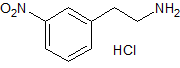
Mirabegron Impurity 21
- Product Number CT-01110-81
- Parent Drug Mirabegron
- CAS Number 19008-62-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Lenalidomide Impurity 16
- Product Number CT-01110-1059
- Parent Drug Lenalidomide
- CAS Number 98475-07-1
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
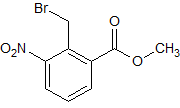
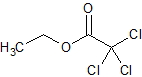
Tadalafil Impurity 50 (Ethyl trichloroacetate)
- Product Number CT-01110-274
- Parent Drug Tadalafil
- CAS Number 515-84-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Amlodipine Impurity 30
- Product Number CT-01110-327
- Parent Drug Amlodipine
- CAS Number 496024-43-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Lansoprazole Impurity 10
- Product Number CT-01110-381
- Parent Drug Lansoprazole
- CAS Number 1083100-27-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Lenvatinib Impurity 15
- Product Number CT-01110-433
- Parent Drug Lenvatinib
- CAS Number 796848-80-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
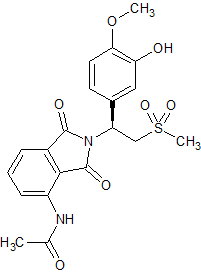
Fosaprepitant Impurity 18
- Product Number CT-01110-490
- Parent Drug Aprepitant
- CAS Number 252742-72-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options