Drug Impurities Reference Standards
Showing 1191–1200 of 1775 results
-
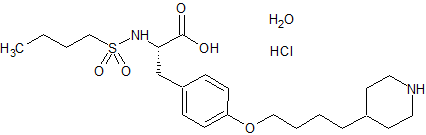
Tirofiban hydrochloride
- Product Number CT-01110-600
- Parent Drug Tirofiban
- CAS Number 150915-40-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
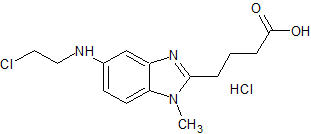
Bendamustine Impurity 8
- Product Number CT-01110-614
- Parent Drug Bendamustine
- CAS Number 1797881-48-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
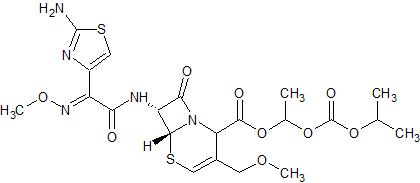
Cefpodoxime Proxetil EP Impurity D
- Product Number CT-01110-877
- Parent Drug Cefpodoxime
- CAS Number 947692-13-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
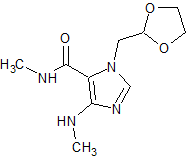
Doxofylline Impurity 1
- Product Number CT-01110-862
- Parent Drug Doxofylline
- CAS Number 1429636-74-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
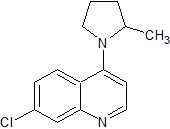
Fasudil Impurity 9
- Product Number CT-01110-678
- Parent Drug Fasudil
- CAS Number 1350827-92-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
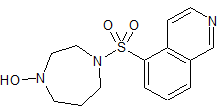
Hydroxychloroquine EP Impurity F
- Product Number CT-01110-1023
- Parent Drug Hydroxychloroquine
- CAS Number 6281-58-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Letrozole Impurity 5
- Product Number CT-01110-1011
- Parent Drug Letrozole
- CAS Number 32446-66-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
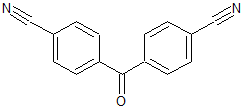
Metformin EP Impurity D
- Product Number CT-01110-548
- Parent Drug Metformin
- CAS Number 108-78-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
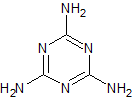
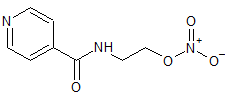
Nicorandil EP Impurity A
- Product Number CT-01110-1063
- Parent Drug Nicorandil
- CAS Number 65141-47-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
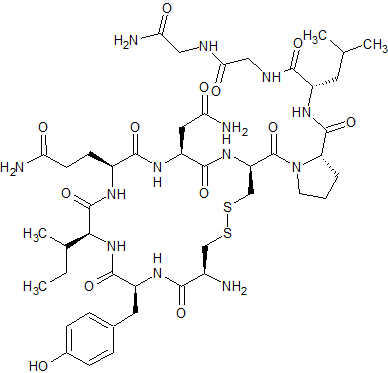
Di-Gly9-Oxytocin
- Product Number CT-01110-1110
- Parent Drug Oxytocin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options