Drug Impurities Reference Standards
Showing 1251–1260 of 1775 results
-
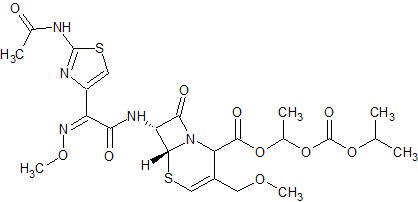
Cefpodoxime Proxetil EP Impurity G
- Product Number CT-01110-880
- Parent Drug Cefpodoxime
- CAS Number 947692-15-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Doxofylline Impurity 4
- Product Number CT-01110-865
- Parent Drug Doxofylline
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
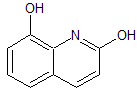
Flumazenil EP Impurity D
- Product Number CT-01110-787
- Parent Drug Flumazenil
- CAS Number 78755-80-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Indacaterol Impurity 4
- Product Number CT-01110-852
- Parent Drug Indacaterol
- CAS Number 15450-76-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
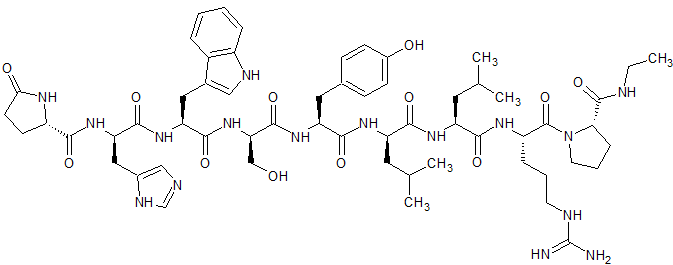
Leuprorelin EP Impurity F
- Product Number CT-01110-1003
- Parent Drug Leuprorelin
- CAS Number 1872435-00-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
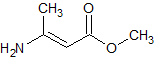
Methyldopa impurity 2
- Product Number CT-01110-938
- Parent Drug Methyldopa
- CAS Number 94-13-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
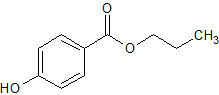
Nifedipine EP Impurity D
- Product Number CT-01110-764
- Parent Drug Nifedipine
- CAS Number 14205-39-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
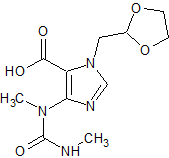
Ozagrel Impurity 1
- Product Number CT-01110-1093
- Parent Drug Ozagrel
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
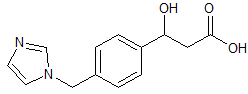
Pranoprofen Impurity 5
- Product Number CT-01110-999
- Parent Drug Pranoprofen
- CAS Number 145986-74-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
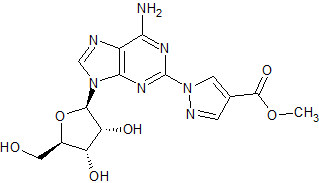
Regadenoson Impurity 3
- Product Number CT-01110-1097
- Parent Drug Regadenoson
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options