Drug Impurities Reference Standards
Showing 171–180 of 1775 results
-
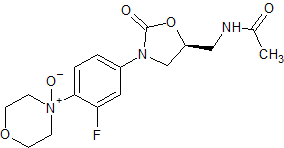
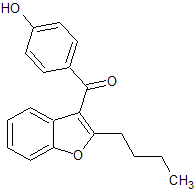
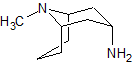
Linezolid Impurity 1
- Product Number CT-01110-166
- Parent Drug Linezolid
- CAS Number 189038-36-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
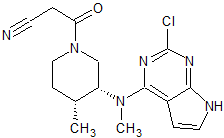
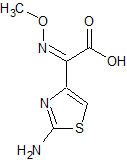
Tofacitinib Impurity 7
- Product Number CT-01110-245
- Parent Drug Tofacitinib
- CAS Number 1616761-00-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
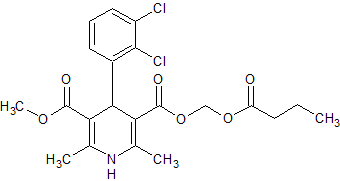
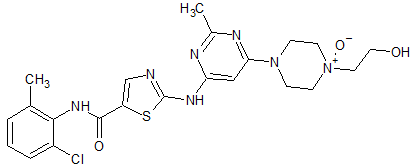
Amlodipine Besylate EP Impurity A
- Product Number CT-01110-323
- Parent Drug Amlodipine
- CAS Number 88150-62-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
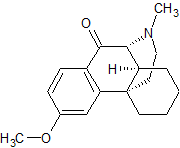
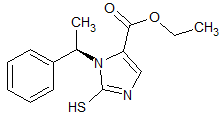
Clevidipine butyrate Impurity 4
- Product Number CT-01110-39
- Parent Drug Clevidipine
- CAS Number 188649-48-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Dextromethorphan EP Impurity C
- Product Number CT-01110-458
- Parent Drug Dextromethorphan
- CAS Number 57969-05-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Amiodarone EP Impurity E
- Product Number CT-01110-778
- Parent Drug Amiodarone
- CAS Number 52490-15-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Cefepime EP Impurity D
- Product Number CT-01110-516
- Parent Drug Cefepime
- CAS Number 65872-41-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Dasatinib Impurity 2
- Product Number CT-01110-891
- Parent Drug Dasatinib
- CAS Number 910297-52-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Etomidate Impurity5
- Product Number CT-01110-1057
- Parent Drug Etomidate
- CAS Number 84711-26-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Granisetron EP Impurity E
- Product Number CT-01110-1091
- Parent Drug Granisetron
- CAS Number 76272-56-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options