Drug Impurities Reference Standards
Showing 211–220 of 1775 results
-
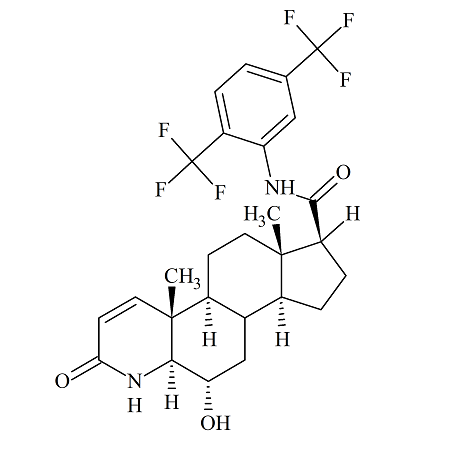
6a-Hydroxy Dutasteride
- Product Number ACA-160819-0021
- Parent Drug Dutasteride
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 4 to 6 week(s)See more size options -
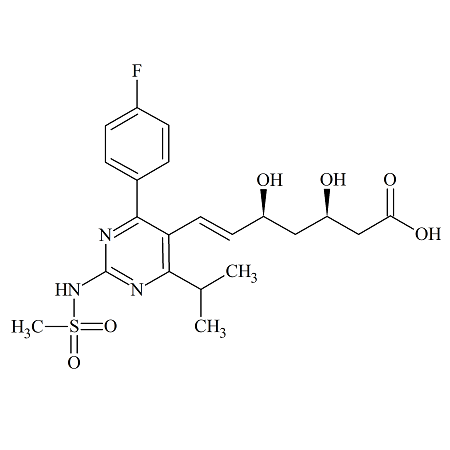
Rosuvastatin N-Desmethyl
- Product Number ROS-12-002
- Parent Drug Rosuvastatin
- CAS Number 371775-74-5
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 4 to 6 week(s)See more size options -
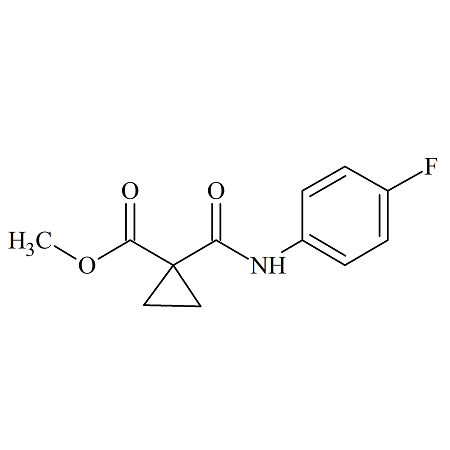
Cabozantinib 4-Fluorocarbamoyl Methyl Ester Impurity
- Product Number B-70831-8
- Parent Drug Cabozantinib
- CAS Number 1345847-71-3
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Adapalene Related Compound A
- Product Number A-10901-01
- Parent Drug Adapalene
- CAS Number 33626-98-1
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
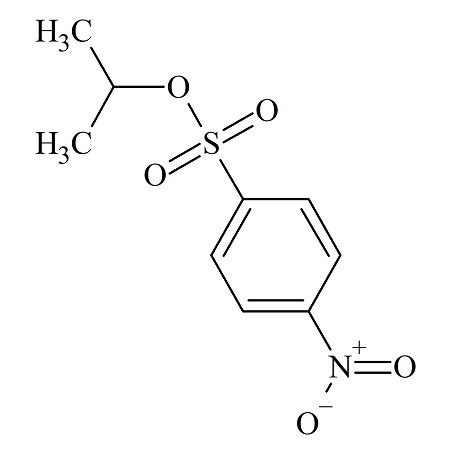
Isopropyl 4-Nitrobenzenesulfonate
- Product Number NBS-10-004
- Parent Drug p-Nitrobenzenesulfonates
- CAS Number 1830-67-7
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 4 to 6 week(s)See more size options -
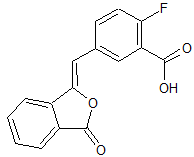
Olaparib Phthalein Despiperazine
- Product Number O-91001-1
- Parent Drug Olaparib
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
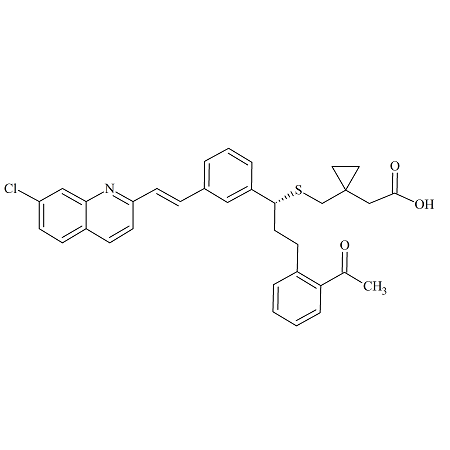
Montelukast Methylketone
- Product Number MON-08-009
- Parent Drug Montelukast
- CAS Number 937275-23-5
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 4 to 6 week(s)See more size options -
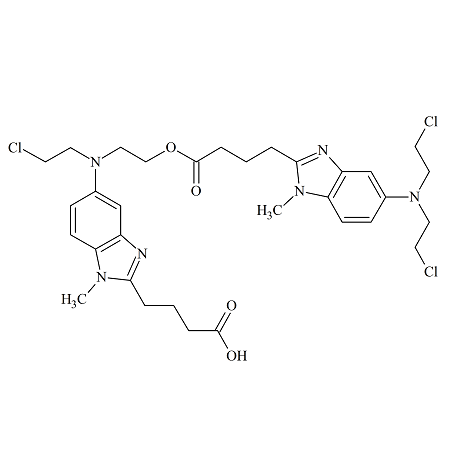
Sorafenib EP Impurity H
- Product Number S-90507-08
- Parent Drug Sorafenib
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 4 to 6 week(s)See more size options