Drug Impurities Reference Standards
Showing 761–770 of 1775 results
-
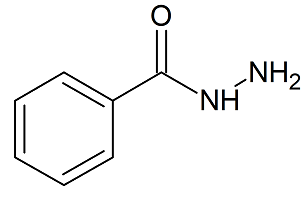
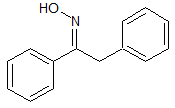
Benzohydrazide
- Product Number ACA-161219-0002
- Parent Drug Azelastine
- CAS Number 613-94-5
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
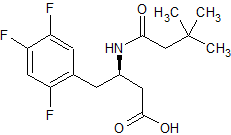
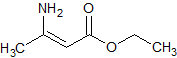
Tadalafil Impurity 39
- Product Number CT-01110-269
- Parent Drug Tadalafil
- CAS Number 14907-27-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Ticagrelor Impurity 44
- Product Number CT-01110-317
- Parent Drug Ticagrelor
- CAS Number 145783-15-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Gefitinib Impurity 13
- Product Number CT-01110-396
- Parent Drug Gefitinib
- CAS Number 95-76-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Sitagliptin Impurity 28
- Product Number CT-01110-440
- Parent Drug Sitagliptin
- CAS Number 486460-00-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
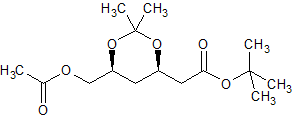
Rosuvastatin Impurity 63
- Product Number CT-01110-499
- Parent Drug Rosuvastatin
- CAS Number 154026-95-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
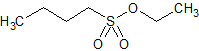
Tirofiban Impurity 22
- Product Number CT-01110-602
- Parent Drug Tirofiban
- CAS Number 2374-68-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
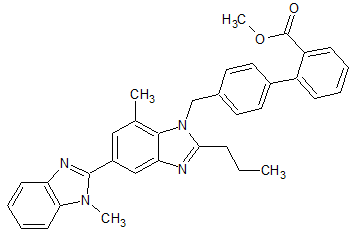
Telmisartan Impurity 30
- Product Number CT-01110-670
- Parent Drug Telmisartan
- CAS Number 1338830-37-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Nifedipine Impurity 11
- Product Number CT-01110-763
- Parent Drug Nifedipine
- CAS Number 626-34-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Parecoxib sodium Impurity 20
- Product Number CT-01110-885
- Parent Drug Parecoxib
- CAS Number 952-06-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options