Drug Impurities Reference Standards
Showing 1501–1510 of 1775 results
-
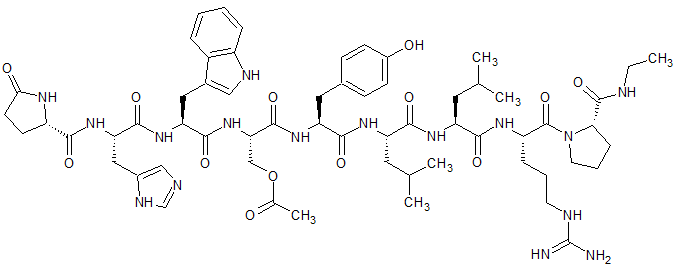
Leuprorelin EP Impurity D
- Product Number CT-01110-1001
- Parent Drug Leuprorelin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
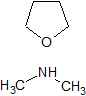
Metformin EP Impurity F
- Product Number CT-01110-549
- Parent Drug Metformin
- CAS Number 124-40-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Nicorandil EP Impurity B
- Product Number CT-01110-1064
- Parent Drug Nicorandil
- CAS Number 6265-73-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
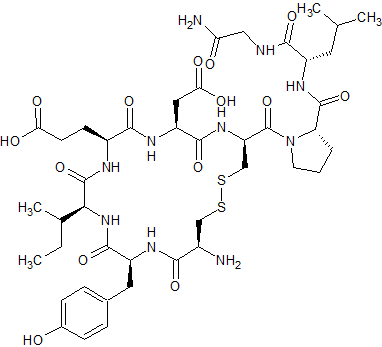
Glu4,Asp5-Oxytocin
- Product Number CT-01110-1112
- Parent Drug Oxytocin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
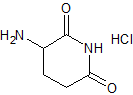
Pomalidomide Impurity 6
- Product Number CT-01110-655
- Parent Drug Pomalidomide
- CAS Number 24666-56-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
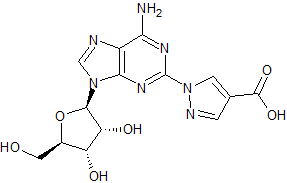
Regadenoson Impurity 1
- Product Number CT-01110-1095
- Parent Drug Regadenoson
- CAS Number 313348-29-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Telmisartan EP Impurity A
- Product Number CT-01110-661
- Parent Drug Telmisartan
- CAS Number 152628-02-9
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
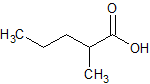
Sodium Valproate EP Impurity L
- Product Number CT-01110-485
- Parent Drug Valproic Acid
- CAS Number 97-61-0
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Tedizolid Impurity 42
- Product Number CT-01110-123
- Parent Drug Tedizolid
- CAS Number 2095200-69-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Linezolid Impurity 26
- Product Number CT-01110-171
- Parent Drug Linezolid
- CAS Number 168828-89-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options