Drug Impurities Reference Standards
Showing 1521–1530 of 1775 results
-
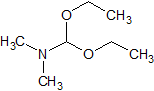
Flumazenil EP Impurity C
- Product Number CT-01110-786
- Parent Drug Flumazenil
- CAS Number 1188-33-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Hydroxychloroquine Impurity 7
- Product Number CT-01110-1024
- Parent Drug Hydroxychloroquine
- CAS Number 47493-14-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
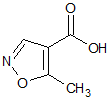
Leflunomide EP Impurity D
- Product Number CT-01110-912
- Parent Drug Leflunomide
- CAS Number 42831-50-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
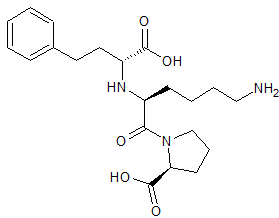
Lisinopril EP Impurity E
- Product Number CT-01110-934
- Parent Drug Lisinopril
- CAS Number 85955-59-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
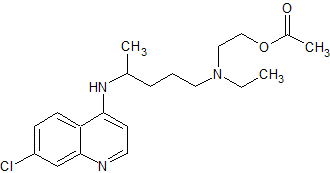
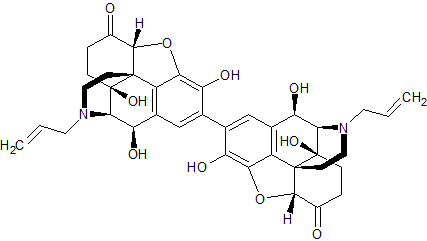
Naloxone Hydrochloride EP Impurity E
- Product Number CT-01110-843
- Parent Drug Naloxone
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
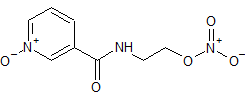
Nicorandil N-Oxide
- Product Number CT-01110-1065
- Parent Drug Nicorandil
- CAS Number 107833-98-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
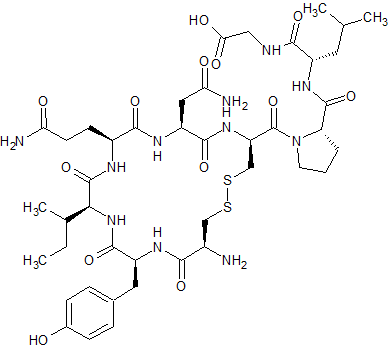
Gly-OH9-Oxytocin
- Product Number CT-01110-1113
- Parent Drug Oxytocin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Pomalidomide Impurity 7
- Product Number CT-01110-656
- Parent Drug Pomalidomide
- CAS Number 5434-21-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
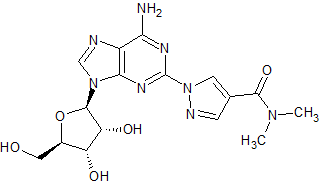
Regadenoson Impurity 2
- Product Number CT-01110-1096
- Parent Drug Regadenoson
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
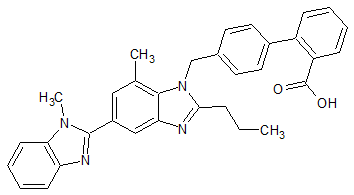
Telmisartan EP Impurity B
- Product Number CT-01110-665
- Parent Drug Telmisartan
- CAS Number 1026353-20-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options