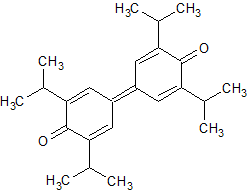
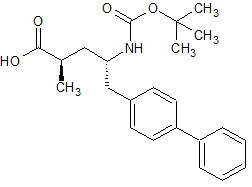
Drug Impurities Reference Standards
Showing 1711–1720 of 1775 results
-
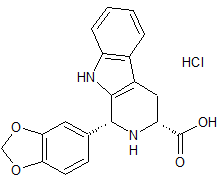
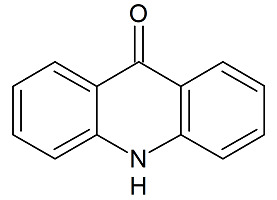
Tadalafil Impurity 29
- Product Number CT-01110-264
- Parent Drug Tadalafil
- CAS Number 474668-76-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
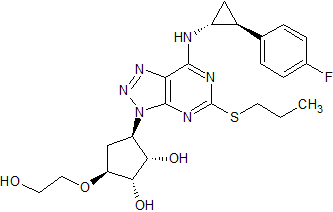
Ticagrelor Impurity 14
- Product Number CT-01110-312
- Parent Drug Ticagrelor
- CAS Number 1643378-47-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
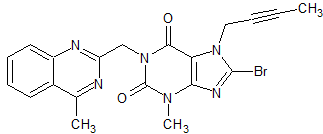
Linagliptin Impurity 48
- Product Number CT-01110-361
- Parent Drug Linagliptin
- CAS Number 853029-57-9
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
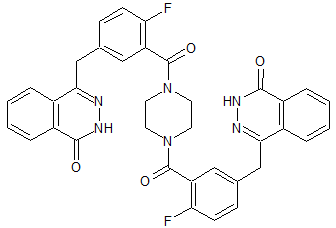
Olaparib Impurity 11
- Product Number CT-01110-421
- Parent Drug Olaparib
- CAS Number 2250242-62-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
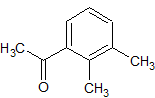
Oxcarbazepine Impurity 17
- Product Number CT-01110-475
- Parent Drug Oxcarbazepine
- CAS Number 578-95-0
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Dexmedetomidine Impurity 31
- Product Number CT-01110-553
- Parent Drug Dexmedetomidine
- CAS Number 2142-71-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
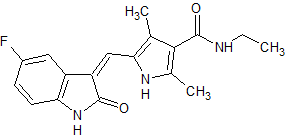
Sunitinib Impurity 24
- Product Number CT-01110-645
- Parent Drug Sunitinib
- CAS Number 1467015-10-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
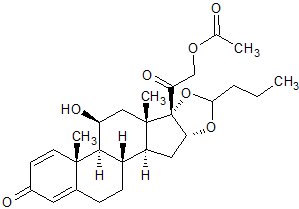
Budesonide EP Impurity K
- Product Number CT-01110-719
- Parent Drug Budesonide
- CAS Number 51333-05-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Propofol Impurity 20
- Product Number CT-01110-827
- Parent Drug Propofol
- CAS Number 2178-51-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
LCZ-696 Impurity 17(S,S)
- Product Number CT-01110-941
- Parent Drug LCZ-696
- CAS Number 1012341-52-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options