Drug Impurities Reference Standards
Showing 1741–1750 of 1775 results
-
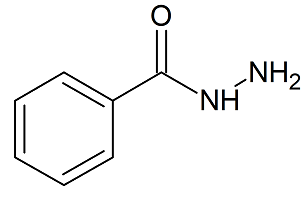
Benzohydrazide
- Product Number ACA-161219-0002
- Parent Drug Azelastine
- CAS Number 613-94-5
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Tadalafil Impurity 39
- Product Number CT-01110-269
- Parent Drug Tadalafil
- CAS Number 14907-27-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Ticagrelor Impurity 44
- Product Number CT-01110-317
- Parent Drug Ticagrelor
- CAS Number 145783-15-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Pramipexole Impurity 11 (and enantiomer)
- Product Number CT-01110-372
- Parent Drug Pramipexole
- CAS Number 1246818-51-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Mesalazine EP Impurity L
- Product Number ACA-161220-0008
- Parent Drug Mesalamine
- CAS Number 118-91-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
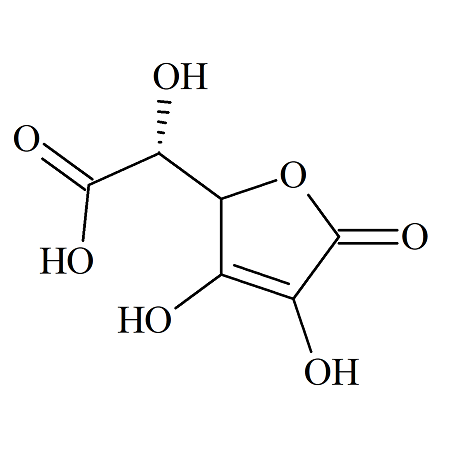
Ascorbic Acid EP Impurity G
- Product Number ASC-16-001
- Parent Drug Ascorbic Acid
- CAS Number 66757-69-5
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
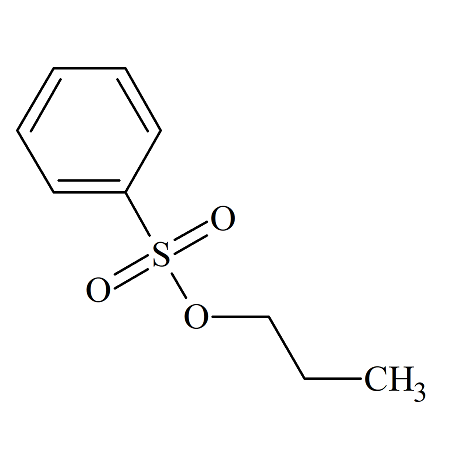
n-Propyl Benzenesulfonate
- Product Number BES-09-001
- Parent Drug Benzenesulfonates
- CAS Number 80-42-2
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
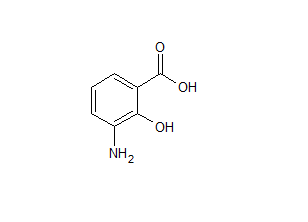
Mesalazine EP Impurity G
- Parent Drug Mesalamine
- CAS Number 490-79-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
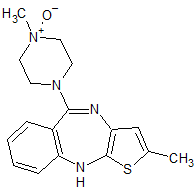
Olanzapine Related Compound C
- Product Number O-10208-02
- Parent Drug Olanzapine
- CAS Number 174794-02-6
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options