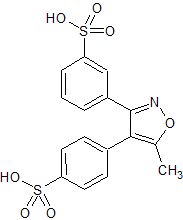
Drug Impurities Reference Standards
Showing 411–420 of 1775 results
-
(4-9)-Oxytocin
- Product Number CT-01110-1106
- Parent Drug Oxytocin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
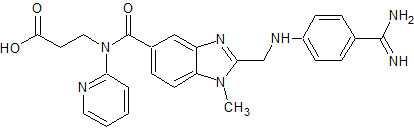
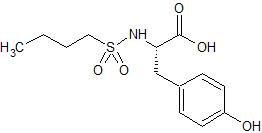
Dabigatran Impurity 21
- Product Number CT-01110-288
- Parent Drug Dabigatran
- CAS Number 211914-51-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
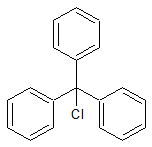
Olmesartan Impurity 29
- Product Number CT-01110-340
- Parent Drug Olmesartan
- CAS Number 76-83-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
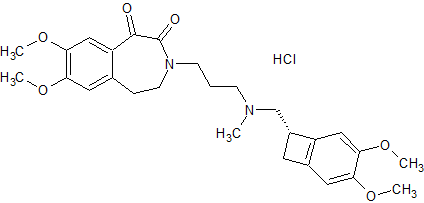
Ivabradine Impurity 10
- Product Number CT-01110-397
- Parent Drug Ivabradine
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
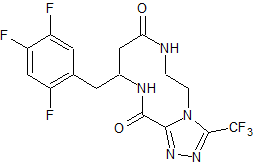
Sitagliptin impurity 29
- Product Number CT-01110-441
- Parent Drug Sitagliptin
- CAS Number 2088771-61-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Rosuvastatin Impurity 65
- Product Number CT-01110-500
- Parent Drug Rosuvastatin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
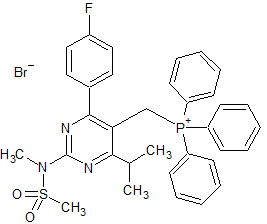
Tirofiban Impurity 23
- Product Number CT-01110-603
- Parent Drug Tirofiban
- CAS Number 149490-60-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
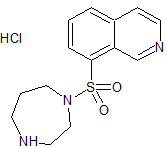
Fasudil Impurity 10
- Product Number CT-01110-673
- Parent Drug Fasudil
- CAS Number 166895-76-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
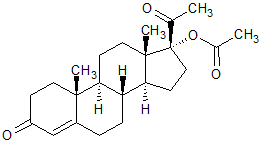
Megestrol Acetate EP Impurity K
- Product Number CT-01110-770
- Parent Drug Megestrol Acetate
- CAS Number 302-23-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Parecoxib Sodium Impurity 26
- Product Number CT-01110-886
- Parent Drug Parecoxib
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options