Drug Impurities Reference Standards
Showing 431–440 of 1775 results
-
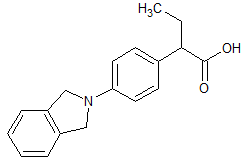
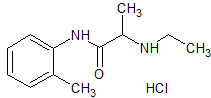
Indobufen Impurity 6
- Product Number CT-01110-984
- Parent Drug Indobufen
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
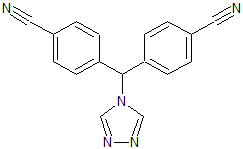
Letrozole EP Impurity A
- Product Number CT-01110-1006
- Parent Drug Letrozole
- CAS Number 112809-52-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
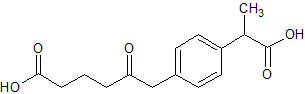
Loxoprofen Impurity 3
- Product Number CT-01110-838
- Parent Drug Loxoprofen
- CAS Number 1091621-61-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
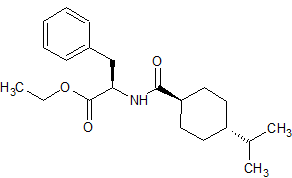
Nateglinide EP Impurity G
- Product Number CT-01110-627
- Parent Drug Nateglinide
- CAS Number 187728-85-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Beta-Asp5-Oxytocin
- Product Number CT-01110-1114
- Parent Drug Oxytocin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Prilocaine EP Impurity C
- Product Number CT-01110-1083
- Parent Drug Prilocaine
- CAS Number 35891-75-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Ribavirin EP Impurity F
- Product Number CT-01110-769
- Parent Drug Ribavirin
- CAS Number 58151-87-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
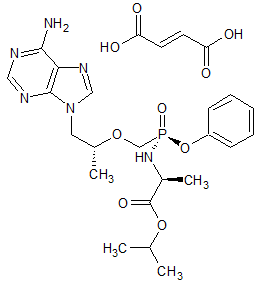
Tenofovir Alafenamide Impurity 3
- Product Number CT-01110-992
- Parent Drug Tenofovir
- CAS Number 1422343-76-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
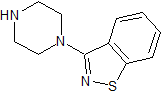
Ziprasidone EP Impurity A
- Product Number CT-01110-574
- Parent Drug Ziprasidone
- CAS Number 87691-87-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
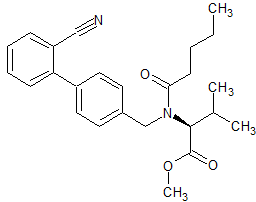
Valsartan ImpurIty 14
- Product Number CT-01110-132
- Parent Drug Valsartan
- CAS Number 137863-90-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options