Drug Impurities Reference Standards
Showing 671–680 of 1775 results
-
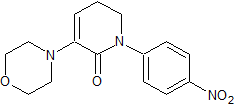
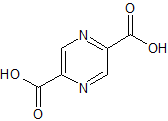
Apixaban Impurity 19
- Product Number CT-01110-277
- Parent Drug Apixaban
- CAS Number 503615-03-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
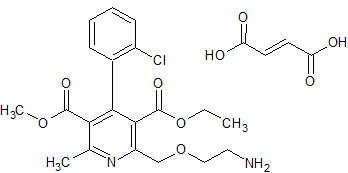
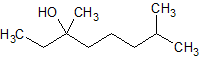
Amlodipine Besylate EP Impurity D Fumarate
- Product Number CT-01110-328
- Parent Drug Amlodipine
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
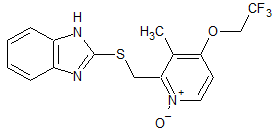
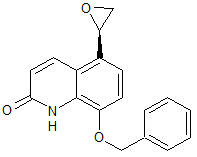
Lansoprazole Impurity 15
- Product Number CT-01110-382
- Parent Drug Lansoprazole
- CAS Number 163119-30-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
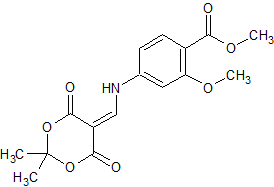
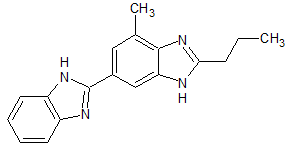
Lenvatinib Impurity 32
- Product Number CT-01110-434
- Parent Drug Lenvatinib
- CAS Number 205448-64-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Aprepitant Impurity 25
- Product Number CT-01110-491
- Parent Drug Aprepitant
- CAS Number 78-69-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Quetiapine EP Impurity W
- Product Number CT-01110-586
- Parent Drug Quetiapine
- CAS Number 1800608-95-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Telmisartan Impurity 11
- Product Number CT-01110-662
- Parent Drug Telmisartan
- CAS Number 884330-09-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Glipizide Impurity 18
- Product Number CT-01110-749
- Parent Drug Glipizide
- CAS Number 122-05-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Indacaterol Impurity 14
- Product Number CT-01110-849
- Parent Drug Indacaterol
- CAS Number 173140-90-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
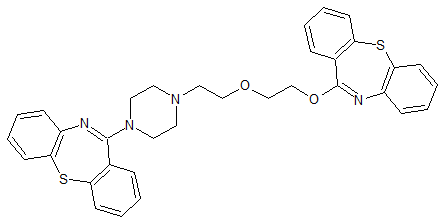
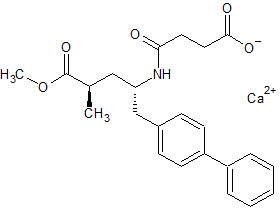
LCZ-696 Impurity 72 calcium salt
- Product Number CT-01110-952
- Parent Drug LCZ-696
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options