Drug Impurities Reference Standards
Showing 681–690 of 1775 results
-
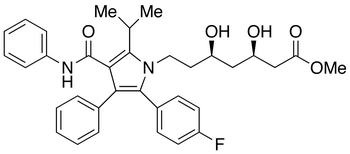
Atorvastatin Methyl Ester
- Product Number ATR-16-004
- Parent Drug Atorvastatin
- CAS Number 345891-62-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Selexipag Impurity A
- Product Number ACB-161114-0004
- Parent Drug Selexipag
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 4 to 6 week(s)See more size options -
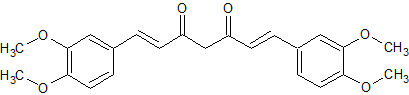
4′,4″-O,O-Dimethylcurcumin
- Product Number C-91127-02
- Parent Drug Curcumin
- CAS Number 52328-98-0
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Alprazolam Impurity 2
- Product Number CT-01110-817
- Parent Drug Alprazolam
- CAS Number 4016-85-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
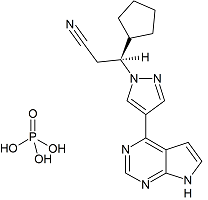
Ruxolitinib Phosphate Impurity A
- Product Number R-30418-01
- Parent Drug Ruxolitinib
- CAS Number NA
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
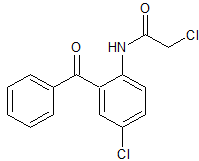
Nintedanib Acid
- Product Number B-71114-0005
- Parent Drug Nintedanib
- CAS Number 894783-71-2
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
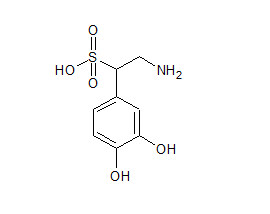
Norepinephrine Sulfonic Acid
- Product Number N-90111-02
- Parent Drug Norepinephrine
- CAS Number 24159-36-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
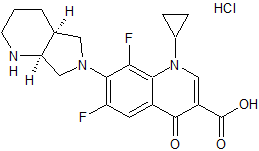
Moxifloxacin EP Impurity A
- Product Number CT-01110-181
- Parent Drug Moxifloxacin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
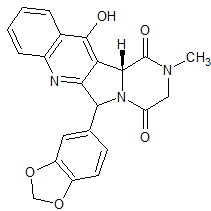
Tadalafil EP Impurity G
- Product Number CT-01110-275
- Parent Drug Tadalafil
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options