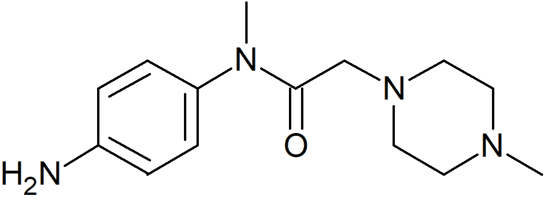Drug Impurities Reference Standards
Showing 801–810 of 1775 results
-
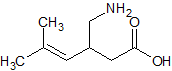
Pregabalin Impurity 12
- Product Number CT-01110-620
- Parent Drug Pregabalin
- CAS Number 216576-74-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
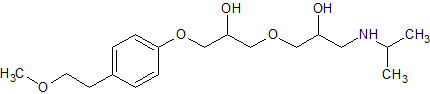
Metoprolol EP Impurity J
- Product Number CT-01110-687
- Parent Drug Metoprolol
- CAS Number 163685-37-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
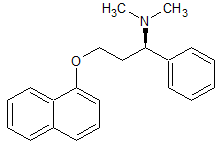
Dapoxetine Impurity 29
- Product Number CT-01110-796
- Parent Drug Dapoxetine
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
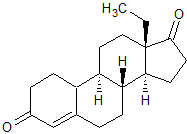
Levonorgestrel EP Impurity L
- Product Number CT-01110-918
- Parent Drug Levonorgestrel
- CAS Number 21800-83-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
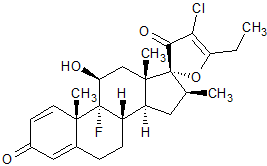
Clobetasol Propionate EP Impurity J
- Product Number C-10426-02
- Parent Drug Clobetasol
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
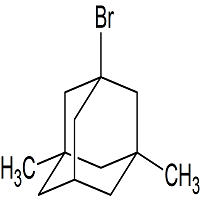
Memantine USP Related Compound D
- Product Number M-11018-01
- Parent Drug Memantine
- CAS Number 941-37-7
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
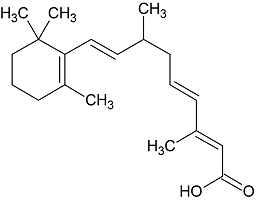
9,10-Dihydroretinoic Acid
- Product Number R-20110-01
- Parent Drug Retinoic Acid
- CAS Number 98299-56-0
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Rivaroxaban Open Ring Acid Impurity
- Product Number R-21214-02
- Parent Drug Rivaroxaban
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
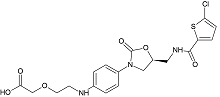
N-(4-Aminophenyl)-N-methyl-2-(4-methylpiperazin-1-yl)acetamide
- Product Number N-40909-01
- Parent Drug Nintedanib
- CAS Number 262368-30-9
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options