Drug Impurities Reference Standards
Showing 831–840 of 1775 results
-
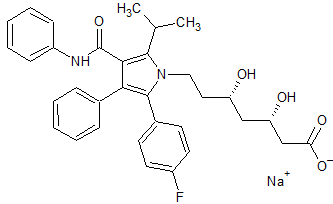
Atorvastatin EP Impurity E(Calcium salt)
- Product Number CT-01110-418
- Parent Drug Atorvastatin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
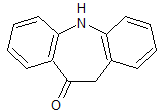
Oxcarbazepine EP Impurity C
- Product Number CT-01110-473
- Parent Drug Oxcarbazepine
- CAS Number 21737-58-6
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Cefradine Impurity 12
- Product Number CT-01110-546
- Parent Drug Cefradine
- CAS Number 26774-89-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Ornidazole Impurity 11
- Product Number CT-01110-637
- Parent Drug Ornidazole
- CAS Number 106-89-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Levofloxacin Impurity 26
- Product Number CT-01110-705
- Parent Drug Levofloxacin
- CAS Number 106939-34-8
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Indometacin EP Impurity F
- Product Number CT-01110-822
- Parent Drug Indometacin
- CAS Number 402849-27-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
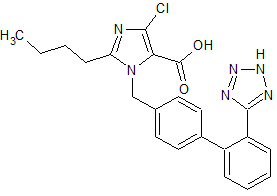
Losartan Impurity 14
- Product Number CT-01110-936
- Parent Drug Losartan
- CAS Number 124750-92-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
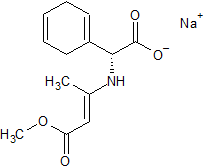
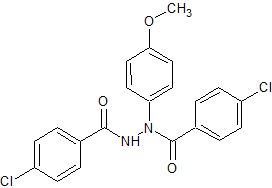
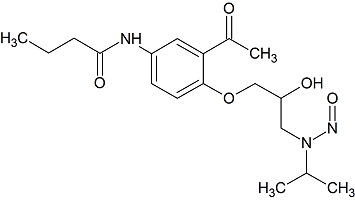
Acebutolol N-nitroso
- Product Number A-10521-01
- Parent Drug Acebutolol
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 3 to 4 week(s)See more size options -
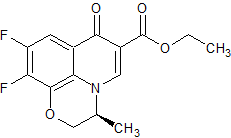
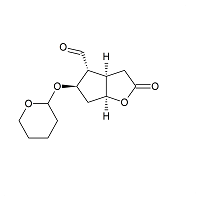
Dehydro Corey Lactone THP
- Product Number L-11029-02
- Parent Drug Lubiprostone
- CAS Number 32233-41-3
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
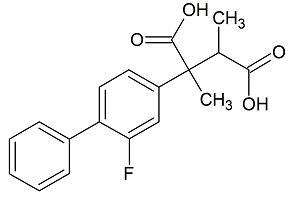
Flurbiprofen EP Impurity B
- Product Number F-20317-01
- Parent Drug Flurbiprofen
- CAS Number 1797883-74-9
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options