Drug Impurities Reference Standards
Showing 911–920 of 1775 results
-
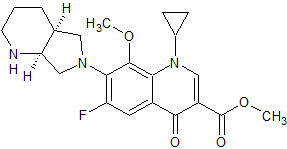
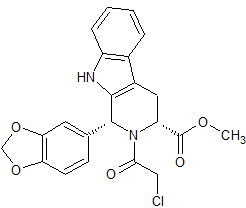
Moxifloxacin Methyl Ester
- Product Number CT-01110-194
- Parent Drug Moxifloxacin
- CAS Number 721970-35-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
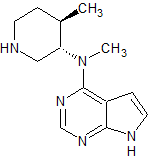
Tofacitinib Impurity 42
- Product Number CT-01110-238
- Parent Drug Tofacitinib
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
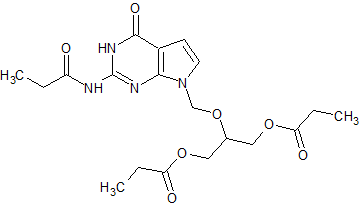
Ganciclovir EP Impurity J
- Product Number CT-01110-71
- Parent Drug Ganciclovir
- CAS Number 177216-32-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
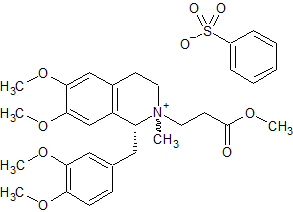
Atracurium Impurity 32
- Product Number CT-01110-1041
- Parent Drug Atracurium
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Tadalafil Impurity 37
- Product Number CT-01110-268
- Parent Drug Tadalafil
- CAS Number 171489-59-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Ticagrelor Impurity 41
- Product Number CT-01110-316
- Parent Drug Ticagrelor
- CAS Number 376608-65-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Linagliptin Impurity 57
- Product Number CT-01110-364
- Parent Drug Linagliptin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
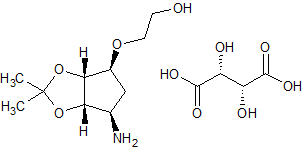
Decitabine Impurity 10
- Product Number CT-01110-425
- Parent Drug Decitabine
- CAS Number 1442660-65-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
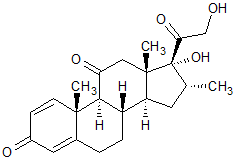
Dexamethasone EP Impurity J
- Product Number CT-01110-478
- Parent Drug Dexamethasone
- CAS Number 2036-77-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
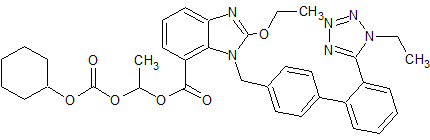
Candesartan Cilexetil EP Impurity E
- Product Number CT-01110-559
- Parent Drug Candesartan
- CAS Number 914613-35-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options