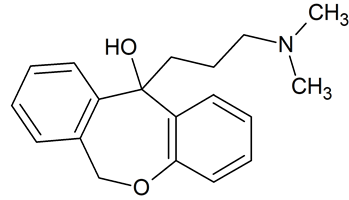Drug Impurities Reference Standards
Showing 941–950 of 1775 results
-
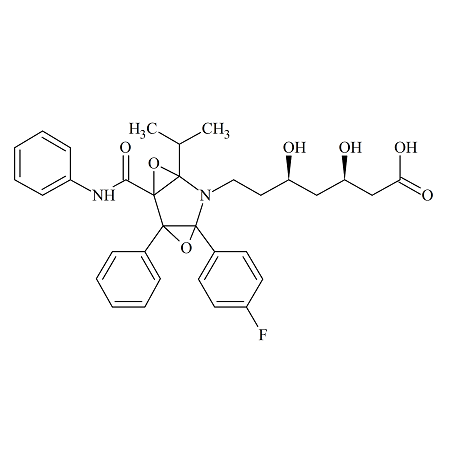
Atorvastatin Diepoxide
- Product Number ATR-16-005
- Parent Drug Atorvastatin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
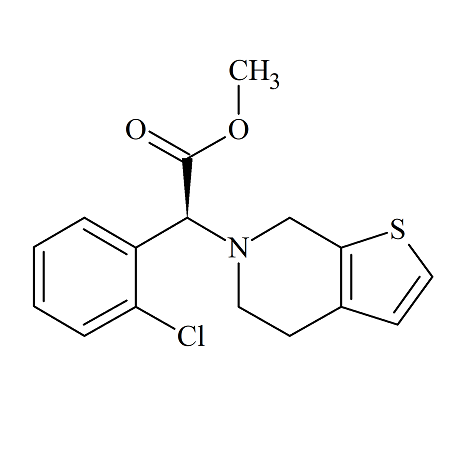
Clopidogrel Impurity 3
- Product Number ACB-161030-0012
- Parent Drug Clopidogrel
- CAS Number 141109-18-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
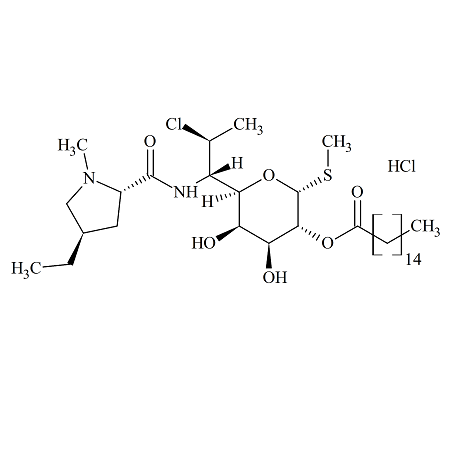
Clindamycin B 2-Palmitate Hydrochloride
- Product Number ACB-170113-0004
- Parent Drug Clindamycin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 4 to 6 week(s)See more size options -
Doxepin Related Compound B
- Product Number D-91223-02
- Parent Drug Doxepin
- CAS Number 4504-88-5
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
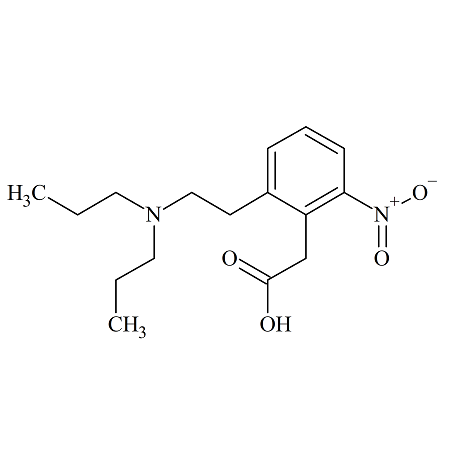
Nitro Ropinirole
- Product Number ROP-10-002
- Parent Drug Ropinirole
- CAS Number 91374-25-3
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 4 to 6 week(s)See more size options -
Afatinib Impurity 1
- Product Number CT-01110-03
- Parent Drug Afatinib
- CAS Number 314771-88-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Monoethyl Felodipine
- Product Number FEL-09-007
- Parent Drug Felodipine
- CAS Number 150131-21-8
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 4 to 6 week(s)See more size options -
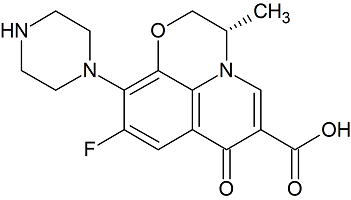
Levofloxacin Related Compound A
- Product Number L-10208-01
- Parent Drug Levofloxacin
- CAS Number 117707-40-1
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options