Drug Impurities Reference Standards
Showing 981–990 of 1775 results
-
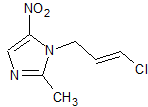
Ornidazole Impurity 8
- Product Number CT-01110-642
- Parent Drug Ornidazole
- CAS Number 1384752-15-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Pimavanserin Impurity 7
- Product Number CT-01110-1090
- Parent Drug Pimavanserin
- CAS Number 1035343
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
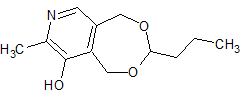
Pyridoxine Impurity 7
- Product Number CT-01110-1049
- Parent Drug Pyridoxine
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Ropivacaine EP Impurity F
- Product Number CT-01110-973
- Parent Drug Ropivacaine
- CAS Number 1945965-95-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
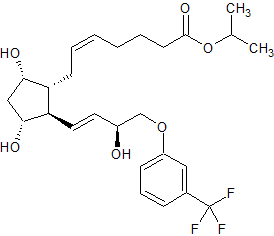
Travoprost Impurity 4
- Product Number CT-01110-1081
- Parent Drug Travoprost
- CAS Number 1420791-14-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
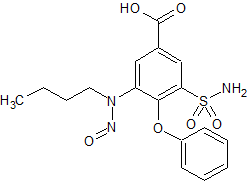
Bumetanide N-Nitroso Impurity
- Product Number B-10312-02
- Parent Drug Bumetanide
- CAS Number 2490432-02-3
- Category Drug Impurities Reference Standards
Temporarily Out of StockSee more size options -
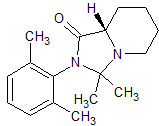
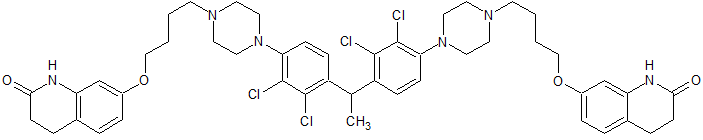
Aripiprazole EP Impurity G
- Product Number CT-01110-15
- Parent Drug Aripiprazole
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
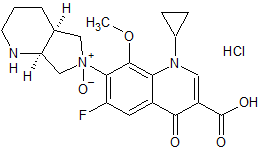
Moxifloxacin Impurity 48
- Product Number CT-01110-199
- Parent Drug Moxifloxacin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
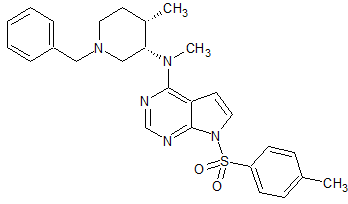
Tofacitinib Impurity 64
- Product Number CT-01110-244
- Parent Drug Tofacitinib
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Mirabegron Impurity 20
- Product Number CT-01110-80
- Parent Drug Mirabegron
- CAS Number 156-28-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options