Drug Impurities Reference Standards
Showing 971–980 of 1775 results
-
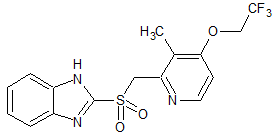
Lansoprazole EP Impurity B/USP Impurity A
- Product Number CT-01110-383
- Parent Drug Lansoprazole
- CAS Number 131926-99-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
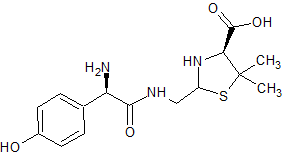
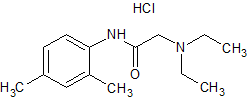
Amoxicillin EP Impurity E
- Product Number CT-01110-452
- Parent Drug Amoxicillin
- CAS Number 1356020-01-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
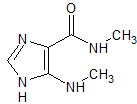
Aminophylline EP Impurity D
- Product Number CT-01110-1047
- Parent Drug Aminophylline
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
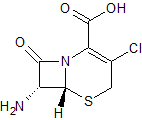
Cefaclor EP Impurity B
- Product Number CT-01110-611
- Parent Drug Cefaclor
- CAS Number 53994-69-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
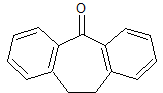
Cyclobenzaprine Impurity 1
- Product Number CT-01110-889
- Parent Drug Cyclobenzaprine
- CAS Number 1210-35-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
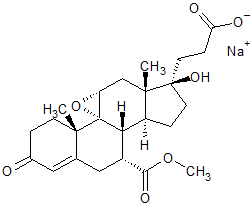
Eplerenone EP Impurity F(Sodium Salt)
- Product Number CT-01110-899
- Parent Drug Eplerenone
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
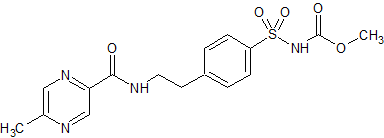
Glipizide EP Impurity G
- Product Number CT-01110-757
- Parent Drug Glipizide
- CAS Number 33288-74-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
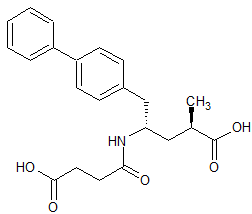
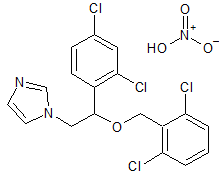
LCZ-696 Impurity 1
- Product Number CT-01110-939
- Parent Drug LCZ-696
- CAS Number 149709-44-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Lidocaine EP Impurity I
- Product Number CT-01110-931
- Parent Drug Lidocaine
- CAS Number 17289-54-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Miconazole EP Impurity D
- Product Number CT-01110-995
- Parent Drug Miconazole
- CAS Number 24168-96-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options