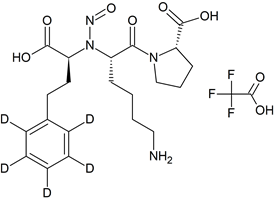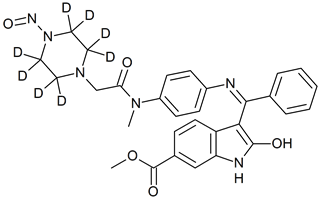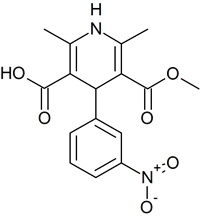Drug Impurities Reference Standards
Showing 1021–1030 of 1775 results
-
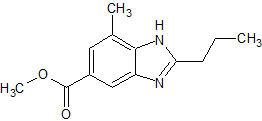
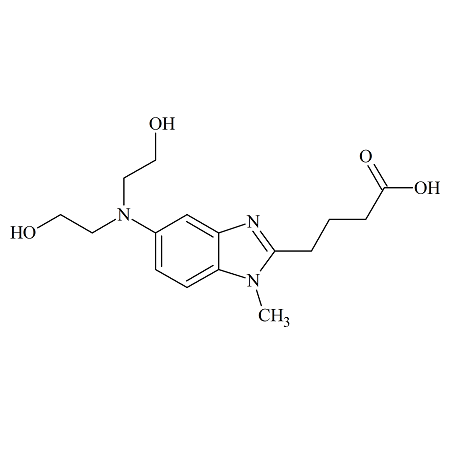
Telmisartan Impurity 13
- Product Number CT-01110-664
- Parent Drug Telmisartan
- CAS Number 152628-00-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Glipizide Impurity 22
- Product Number CT-01110-752
- Parent Drug Glipizide
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Indacaterol Impurity 21
- Product Number CT-01110-851
- Parent Drug Indacaterol
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Oseltamivir EP Impurity F(Mixture of Diastereomers)
- Product Number CT-01110-960
- Parent Drug Oseltamivir
- CAS Number 1052063-37-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
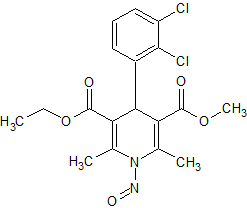
Felodipine N-Nitroso
- Product Number F-10521-01
- Parent Drug Felodipine
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 2 to 3 week(s)See more size options -
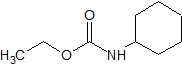
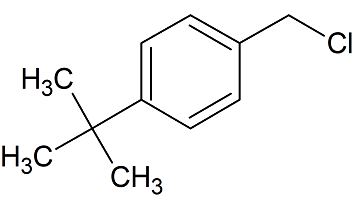
tert-Butylbenzyl Chloride
- Product Number B-11029-08
- Parent Drug Butenafine
- CAS Number 19692-45-6
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
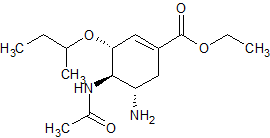
Lisinopril N-Nitroso-D5
- Product Number L-20726-01
- Parent Drug Lisinopril
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
N-Desmethylnintedanib-D8 N-nitroso
- Product Number N-40528-02
- Parent Drug Nintedanib
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Nicardipine USP Related Compound A
- Product Number N-50311-01
- Parent Drug Nicardipine
- CAS Number 74936-72-4
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options