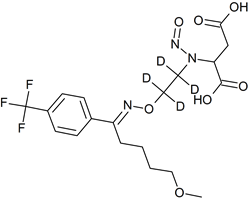Drug Impurities Reference Standards
Showing 1051–1060 of 1775 results
-
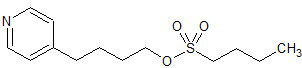
Tirofiban Impurity 29
- Product Number CT-01110-605
- Parent Drug Tirofiban
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
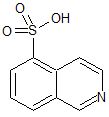
Fasudil Impurity 15
- Product Number CT-01110-675
- Parent Drug Fasudil
- CAS Number 27655-40-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
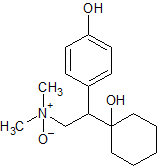
Venlafaxine Impurity 15
- Product Number CT-01110-790
- Parent Drug Venlafaxine
- CAS Number 1021933-95-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
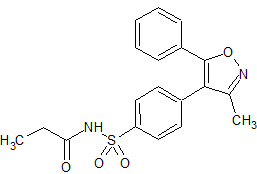
Parecoxib Sodium Impurity 46
- Product Number CT-01110-888
- Parent Drug Parecoxib
- CAS Number 477594-28-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
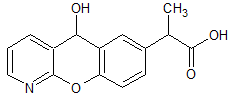
Pranoprofen Impurity 15
- Product Number CT-01110-997
- Parent Drug Pranoprofen
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
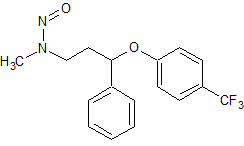
Fluoxetine N-Nitroso
- Product Number F-10521-02
- Parent Drug Fluoxetine
- CAS Number 150494-06-7
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 2 to 3 week(s)See more size options -
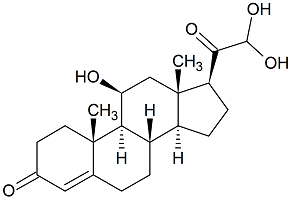
Corticosterone 21-Aldehyde Hydrate
- Product Number C-20104-01
- Parent Drug Corticosterone
- CAS Number 68473-77-8
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
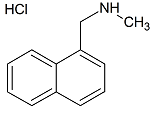
Terbinafine EP Impurity A
- Product Number T-30117-01
- Parent Drug Terbinafine
- CAS Number 65473-13-4
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Fluvoxamine EP Impurity C-D4 N-nitroso
- Product Number F-40820-03
- Parent Drug Fluvoxamine
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options