Drug Impurities Reference Standards
Showing 1031–1040 of 1775 results
-
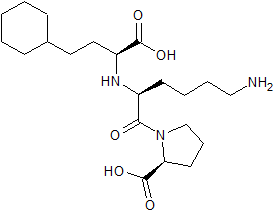
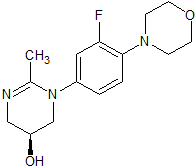
Lisinopril EP Impurity F
- Product Number CT-01110-935
- Parent Drug Lisinopril
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
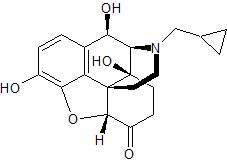
Naltrexone EP Impurity G
- Product Number CT-01110-832
- Parent Drug Naltrexone
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
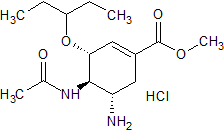
Oseltamivir EP Impurity E(HCl)
- Product Number CT-01110-963
- Parent Drug Oseltamivir
- CAS Number 208720-71-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
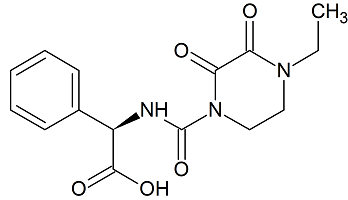
Piperacillin Sodium EP Impurity G
- Product Number CT-01110-968
- Parent Drug Piperacillin
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 2-3 week(s)See more size options -
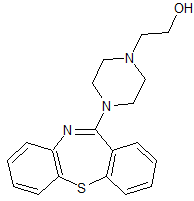
Quetiapine Impurity 2
- Product Number CT-01110-585
- Parent Drug Quetiapine
- CAS Number 329216-67-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
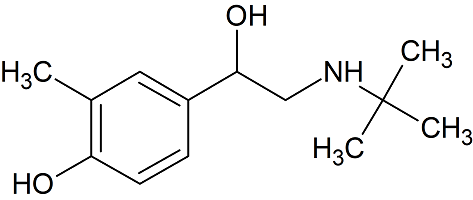
Salbutamol EP Impurity C
- Product Number CT-01110-502
- Parent Drug Salbutamol
- CAS Number 18910-68-4
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 3-4 week(s)See more size options -
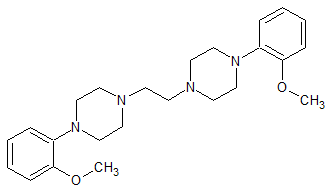
Urapidil Impurity 9
- Product Number CT-01110-1079
- Parent Drug Urapidil
- CAS Number 108997-11-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
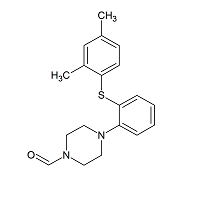
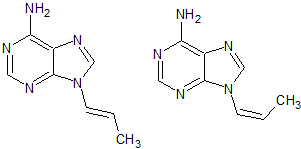
Vortioxetine Impurity 31
- Product Number CT-01110-127
- Parent Drug Vortioxetine
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 3-4 week(s)See more size options -
Linezolid Impurity 30
- Product Number CT-01110-174
- Parent Drug Linezolid
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Tenofovir disoproxil Impurity 32
- Product Number CT-01110-218
- Parent Drug Tenofovir
- CAS Number 4121-40-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options