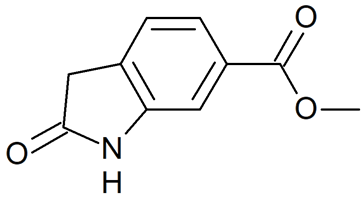Drug Impurities Reference Standards
Showing 1361–1370 of 1775 results
-
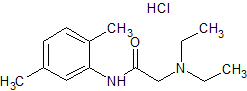
Lidocaine EP Impurity J
- Product Number CT-01110-921
- Parent Drug Lidocaine
- CAS Number 1012864-23-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
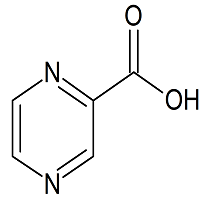
Pyrazinamide EP Impurity A
- Product Number P-11018-01
- Parent Drug Pyrazinamide
- CAS Number 98-97-5
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
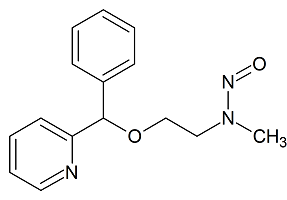
N,C-Didesmethyl Doxylamine N-Nitroso
- Product Number D-20126-01
- Parent Drug Doxylamine
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Desethylsulfonylbaricitinib N-nitroso
- Product Number B-31221-02
- Parent Drug Baricitinib
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Methyl 2-oxoindoline-6-carboxylate
- Product Number N-40909-02
- Parent Drug Nintedanib
- CAS Number 14192-26-8
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
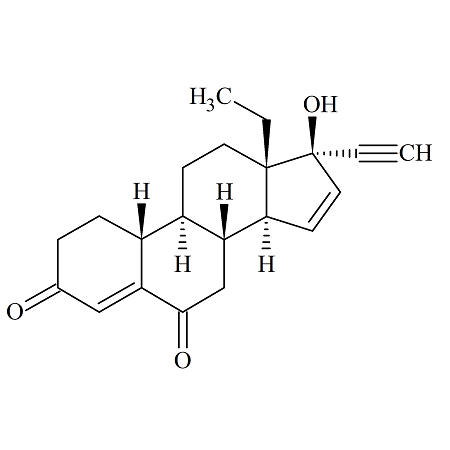
Gestodene Impurity E
- Product Number ACB-161030-0031
- Parent Drug Gestodene
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
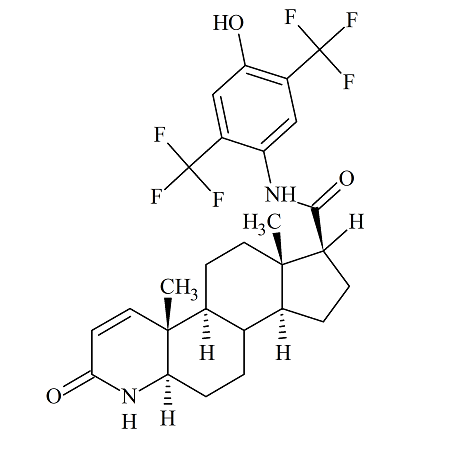
4’-Hydroxy Dutasteride
- Product Number ACA-160819-0022
- Parent Drug Dutasteride
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 4 to 6 week(s)See more size options -
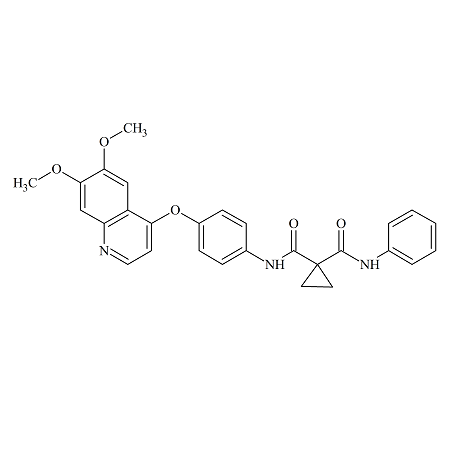
Cabozantinib Desfluoro
- Product Number B-70831-3
- Parent Drug Cabozantinib
- CAS Number 849221-94-9
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
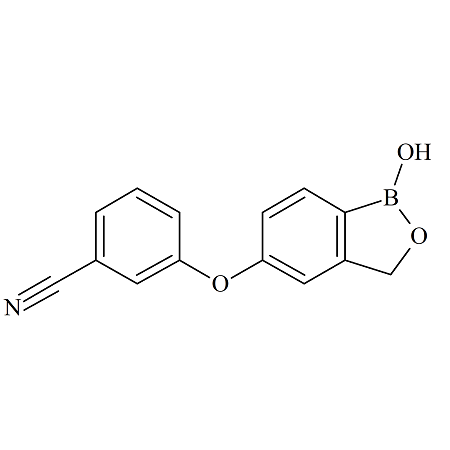
Crisaborole m-Isomer
- Product Number ACB-170828-0003
- Parent Drug Crisaborole
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options