Drug Impurities Reference Standards
Showing 1381–1390 of 1775 results
-
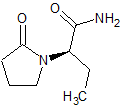
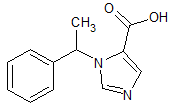
Levetiracetam EP Impurity D
- Product Number CT-01110-161
- Parent Drug Levetiracetam
- CAS Number 103765-01-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
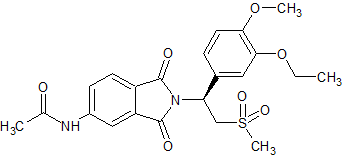
Apremilast Impurity 4
- Product Number CT-01110-24
- Parent Drug Apremilast
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
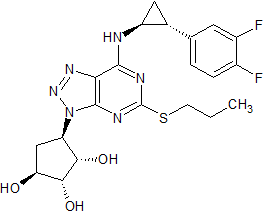
Ticagrelor Impurity 7
- Product Number CT-01110-321
- Parent Drug Ticagrelor
- CAS Number 220347-05-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
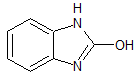
Lansoprazole EP Impurity D(Rabeprazole EP Impurity K)
- Product Number CT-01110-388
- Parent Drug Lansoprazole
- CAS Number 615-16-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
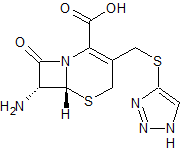
Cefoperazone EP Impurity D
- Product Number CT-01110-454
- Parent Drug Cefoperazone
- CAS Number 37539-03-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
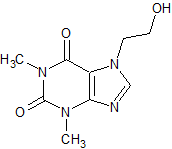
Aminophylline EP ImpurityF
- Product Number CT-01110-1045
- Parent Drug Aminophylline
- CAS Number 519-37-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Cefaclor Impurity 9
- Product Number CT-01110-613
- Parent Drug Cefaclor
- CAS Number 6485-67-2
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
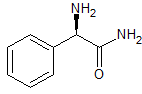
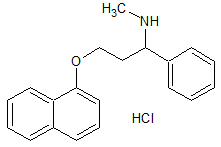
Dapoxetine Impurity 3
- Product Number CT-01110-797
- Parent Drug Dapoxetine
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
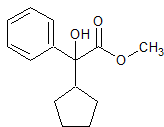
Etomidate EP Impurity A
- Product Number CT-01110-1058
- Parent Drug Etomidate
- CAS Number 3157-27-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Glycopyrrolate Impurity 2
- Product Number CT-01110-783
- Parent Drug Glycopyrrolate
- CAS Number 19833-96-6
- Category Drug Impurities Reference Standards
Pending QCSee more size options