Drug Impurities Reference Standards
Showing 1391–1400 of 1775 results
-
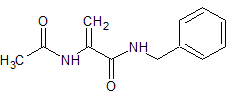
Lacosamide EP Impurity K
- Product Number CT-01110-1017
- Parent Drug Lacosamide
- CAS Number 86921-49-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
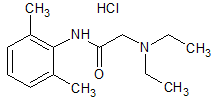
Lidocaine EP Impurity F(Hydrochloride)
- Product Number CT-01110-928
- Parent Drug Lidocaine
- CAS Number 857170-72-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
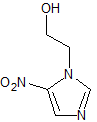
Metronidazole EP Impurity D
- Product Number CT-01110-745
- Parent Drug Metronidazole
- CAS Number 5006-68-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
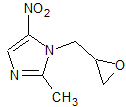
Ornidazole Impurity 3
- Product Number CT-01110-639
- Parent Drug Ornidazole
- CAS Number 16773-52-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
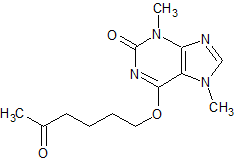
Pentoxifylline EP Impurity G
- Product Number CT-01110-967
- Parent Drug Pentoxifylline
- CAS Number 93079-86-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
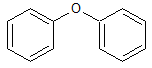
Propofol EP Impurity I
- Product Number CT-01110-831
- Parent Drug Propofol
- CAS Number 101-84-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
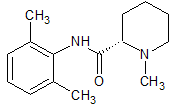
Ropivacaine EP Impurity C
- Product Number CT-01110-975
- Parent Drug Ropivacaine
- CAS Number 24358-84-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
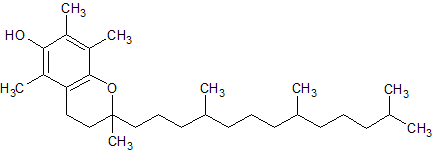
Tocopherol Impurity 5
- Product Number CT-01110-519
- Parent Drug Tocopherol
- CAS Number 10191-41-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
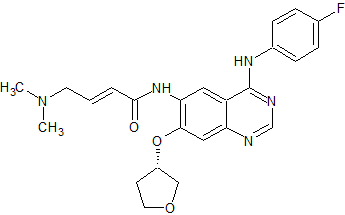
Afatinib Impurity 18
- Product Number CT-01110-05
- Parent Drug Afatinib
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
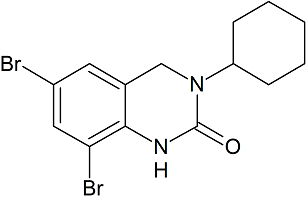
Bromhexine Impurity 11
- Product Number CT-01110-141
- Parent Drug Bromhexine
- CAS Number N/A
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 3-4 week(s)See more size options