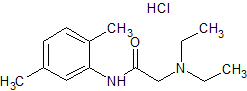
Drug Impurities Reference Standards
Showing 1421–1430 of 1775 results
-
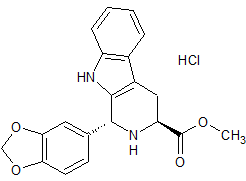
Tadalafil Impurity 12
- Product Number CT-01110-256
- Parent Drug Tadalafil
- CAS Number 171596-44-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
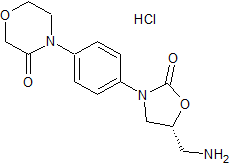
Rivaroxaban Impurity 42
- Product Number CT-01110-301
- Parent Drug Rivaroxaban
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
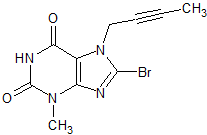
Linagliptin Impurity 31
- Product Number CT-01110-354
- Parent Drug Linagliptin
- CAS Number 666816-98-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
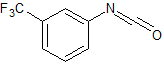
Sorafenib Impurity 20
- Product Number CT-01110-411
- Parent Drug Sorafenib
- CAS Number 329-01-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
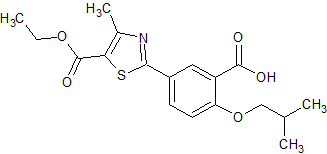
Febuxostat Impurity 58
- Product Number CT-01110-464
- Parent Drug Febuxostat
- CAS Number 2095166-41-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
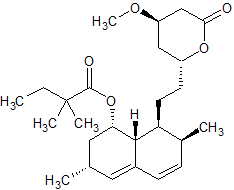
Simvastatin Impurity 12
- Product Number CT-01110-529
- Parent Drug Simvastatin
- CAS Number 864357-88-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
(R)-Pregabalin
- Product Number CT-01110-623
- Parent Drug Pregabalin
- CAS Number 148553-51-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
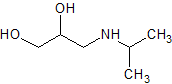
Metoprolol EP Impurity N
- Product Number CT-01110-689
- Parent Drug Metoprolol
- CAS Number 6452-57-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
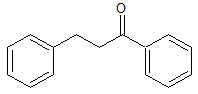
Dapoxetine Impurity 34
- Product Number CT-01110-799
- Parent Drug Dapoxetine
- CAS Number 1083-30-3
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Lidocaine EP Impurity J
- Product Number CT-01110-921
- Parent Drug Lidocaine
- CAS Number 1012864-23-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options