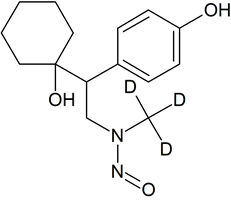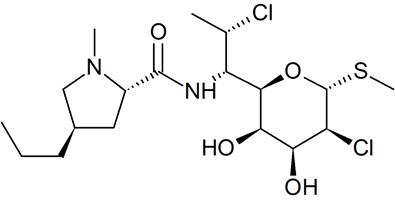Drug Impurities Reference Standards
Showing 1411–1420 of 1775 results
-
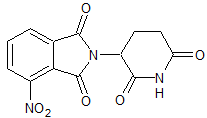
Pomalidomide Impurity 11
- Product Number CT-01110-651
- Parent Drug Pomalidomide
- CAS Number 19171-18-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
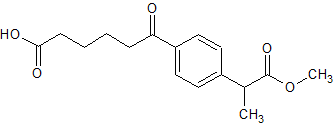
Pantoprazole Impurity 36
- Product Number CT-01110-733
- Parent Drug Pantoprazole
- CAS Number 768386-37-4
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
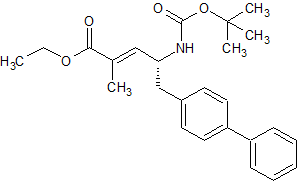
Loxoprofen Impurity 32
- Product Number CT-01110-840
- Parent Drug Loxoprofen
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
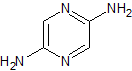
LCZ-696 Impurity 22
- Product Number CT-01110-945
- Parent Drug LCZ-696
- CAS Number 149709-59-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
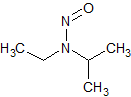
N-nitrosoisopropylethyl amine (NIPEA)
- Product Number N-10521-06
- Parent Drug Nitroso Compounds
- CAS Number 16339-04-1
- Category Drug Impurities Reference Standards
Made to Order - Lead Time: 2 to 3 week(s)See more size options -
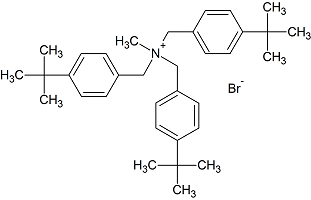
Butenafine Tris(t-butylbenzyl) Bromide
- Product Number B-11029-04
- Parent Drug Butenafine
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Calcipotriol Dimer Mixture
- Product Number C-20329-01
- Parent Drug Calcipotriol
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
N-Desmethyl Desvenlafaxine-D3 N-nitroso
- Product Number V-50826-01
- Parent Drug Desvenlafaxine
- CAS Number N/A
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Clindamycin Impurity 47
- Product Number C-50103-01
- Parent Drug Clindamycin
- CAS Number N/A
- Category Drug Impurities Reference Standards
DiscontinuedSee more size options