Drug Impurities Reference Standards
Showing 1401–1410 of 1775 results
-
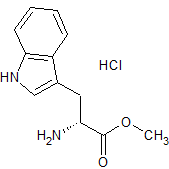
Moxifloxacin Impurity 39
- Product Number CT-01110-195
- Parent Drug Moxifloxacin
- CAS Number 151213-40-0
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Tofacitinib Impurity 50
- Product Number CT-01110-240
- Parent Drug Tofacitinib
- CAS Number 2227199-31-5
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
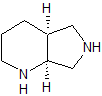
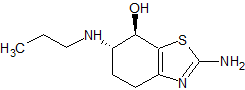
endo-Lurasidone
- Product Number CT-01110-77
- Parent Drug Lurasidone
- CAS Number 1318074-25-7
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
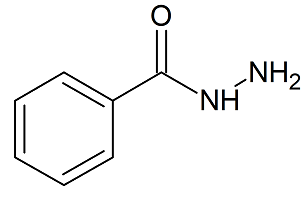
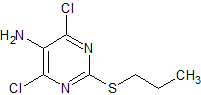
Benzohydrazide
- Product Number ACA-161219-0002
- Parent Drug Azelastine
- CAS Number 613-94-5
- Category Drug Impurities Reference Standards
In Stock - Ready to ShipSee more size options -
Tadalafil Impurity 39
- Product Number CT-01110-269
- Parent Drug Tadalafil
- CAS Number 14907-27-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Ticagrelor Impurity 44
- Product Number CT-01110-317
- Parent Drug Ticagrelor
- CAS Number 145783-15-9
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
Pramipexole Impurity 11 (and enantiomer)
- Product Number CT-01110-372
- Parent Drug Pramipexole
- CAS Number 1246818-51-8
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
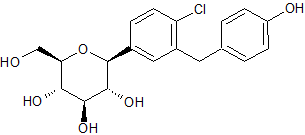
Dapagliflozin Impurity 13
- Product Number CT-01110-427
- Parent Drug Dapagliflozin
- CAS Number 864070-37-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
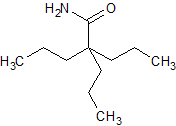
Sodium Valproate EP Impurity G
- Product Number CT-01110-480
- Parent Drug Valproic Acid
- CAS Number 52061-73-1
- Category Drug Impurities Reference Standards
Pending QCSee more size options -
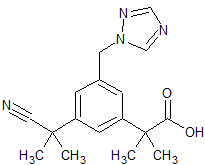
Anastrozole Impurity 14
- Product Number CT-01110-571
- Parent Drug Anastrozole
- CAS Number N/A
- Category Drug Impurities Reference Standards
Pending QCSee more size options